Researchers from the University of Cambridge have this month published the results of a collaborative project, supported by the OpenPlant Fund, to develop droplet-based microfluidics systems for rapid prototyping in plant systems. The paper describes how they have achieved on-chip encapsulation and analysis of protoplasts isolated from the emergent plant model Marchantia polymorpha. We caught up with the team to find out more about their OpenPlant Fund project. Read on to find out more...
Yu Z, Boehm CR, Hibberd JM, Abell C, Haseloff J, Burgess SJ, et al. (2018) Droplet-based microfluidic analysis and screening of single plant cells. PLoS ONE 13(5): e0196810.
Publication abstract
Droplet-based microfluidics has been used to facilitate high-throughput analysis of individual prokaryote and mammalian cells. However, there is a scarcity of similar workflows applicable to rapid phenotyping of plant systems where phenotyping analyses typically are time-consuming and low-throughput. We report on-chip encapsulation and analysis of protoplasts isolated from the emergent plant model Marchantia polymorpha at processing rates of >100,000 cells per hour. We use our microfluidic system to quantify the stochastic properties of a heat-inducible promoter across a population of transgenic protoplasts to demonstrate its potential for assessing gene expression activity in response to environmental conditions. We further demonstrate on-chip sorting of droplets containing YFP-expressing protoplasts from wild type cells using dielectrophoresis force. This work opens the door to droplet-based microfluidic analysis of plant cells for applications ranging from high-throughput characterisation of DNA parts to single-cell genomics to selection of rare plant phenotypes.
Interview with the OpenPlant Fund team
A brief overview of the project
A current limitation for plant synthetic biology involves high-throughput screening of genetic parts in plants. Current approaches require testing circuits in individual plants, through transient or stable transgenics. Applying these techniques to hundreds of different circuits is not feasible at a laboratory scale. In this project, we use droplet based microfluidics to isolate and characterise both gene expression activity and chlorophyll content on single plant protoplasts at a high throughput scale. Our device can potentially analyse protoplasts at a processing rate of > 100,000 cells per hour. We use this system to quantify the stochastic properties of a heat-inducible promoter across a population of transgenic Marchantia polymorpha protoplasts to demonstrate its potential for assessing gene expression activity in response to environmental conditions. In addition, we managed to sort droplets containing YFP-expressing protoplasts from wild type cells using dielectrophoresis force. This work opens the door to droplet-based microfluidic analysis of plant cells for applications ranging from high-throughput characterisation of DNA parts to single-cell genomics to selection of rare plant phenotypes.
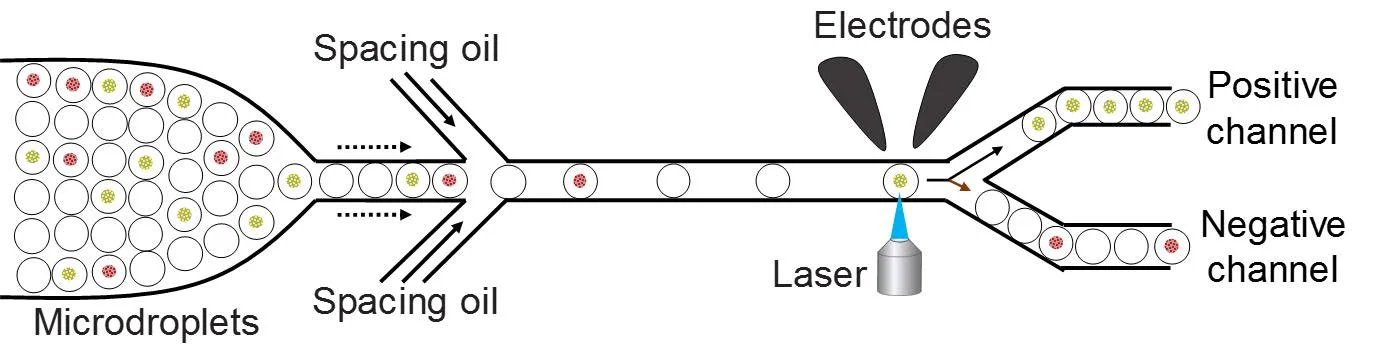
A schematic of the droplet based microfluidics setup
What inspired the project?
Part of our research is focused on identifying DNA regulatory elements that could be used for designing synthetic promoters in plants. To achieve this, we would normally create a reporter construct containing the element of interest and then test its activity on individual plants through transient or stable genetic transformation. This approach can be very time consuming and impedes the researcher to test more than a handful of constructs at the time. Based on this we consider there was a need to develop methods that could accelerate the process. We knew Droplet-based microfluidics has been used to facilitate high throughput analysis of individual prokaryote and mammalian cells so we thought implementing this method in plants will be very useful.
How did this project develop links between Cambridge and Norwich?
During the early stages of the project we teamed up with Oleg Raitskin from the Patron lab at the Earlham Institute. Oleg was very kind to share his experiences with protoplast isolation and also he showed us his method for protoplast transformation in tobacco leaves.
Microscopy images of protoplasts captured in droplets and sorted by fluorescence
What was your favourite aspect of the project?
We really enjoyed learning more about each other’s area of expertise. This project spans the disciplines of physical chemistry optomechatronics and biology so it gave us an opportunity to approach disciplines in which we were not very familiar.
Sorting of M. polymorpha protoplasts: Microscopy images of microdroplets sorted into positive and negative channels based on their fluorescence intensity.
What is the biggest challenge the team faced?
Preparing high quality protoplast preparations from Marchantia was one of the greater challenges we encountered.
Is there something that came out of the project that you never expected at the beginning?
Being able to perform the sorting was unexpected at the beginning. Protoplasts are very sensitive cells that can burst spontaneously. The fact that we manage not just to isolate and measure but also sort opens a lot of possibilities for further applications.
How did the OpenPlant Fund enable the development of the project?
We could have never done this kind of project without the OpenPlant Fund. Apart of the funding which played a primal role in the development of the project, the OpenPlant fund offered great support across the whole process. For instance, our collaboration was established during a fund mixer organized by the synbio fund. Thanks to the Open Plant initiative we showed our project in various Open Plant meetings which resulted in very useful comments from various colleagues. Finally the costs derived from publishing the paper and making it open access were also covered by the grant.
What are the future opportunities to take this project forward?
Now that the system is set up, the next step could be to expand it to on-chip protoplast transformation (as has been done for other cell types). Protoplast transformation currently requires large amounts of materials (cells and DNA) and is low throughput, so this would be a big plus.